BBRC 2022 (Tatsuguchi, Uruno et al.)
Pharmacological intervention of cholesterol sulfate-mediated T cell exclusion promotes antitumor immunity.
Tatsuguchi T, Uruno T, Sugiura Y, Oisaki K, Takaya D, Sakata D, Izumi Y, Togo T, Hattori Y, Kunimura K, Sakurai T, Honma T, Bamba T, Nakamura M, Kanai M, Suematsu M, Fukui Y:
Biochemical and Biophysical Research Communications 609: 183-188, 2022.
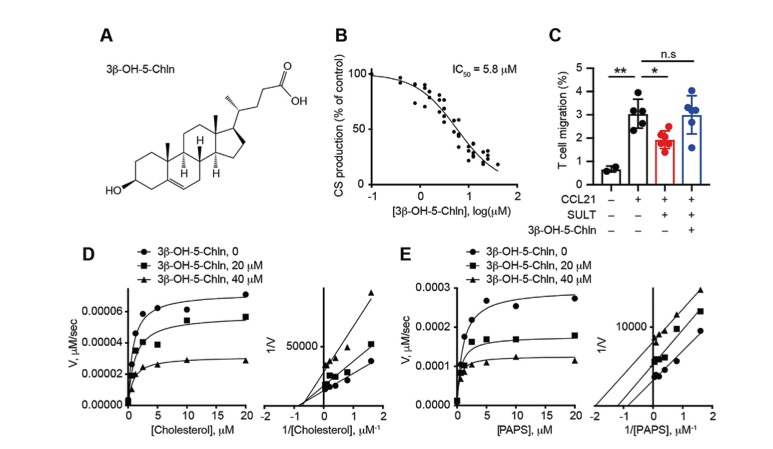
Effective cancer immunotherapy requires physical contact of T cells with cancer cells. However, tumors often constitute special microenvironments that exclude T cells and resist immunotherapy. Cholesterol sulfate (CS) is a product of sulfotransferase SULT2B1b and acts as an endogenous inhibitor of DOCK2, a Rac activator essential for migration and activation of lymphocytes. We have recently shown that cancer-derived CS prevents tumor infiltration by effector T cells. Therefore, SULT2B1b may be a therapeutic target to dampen CS-mediated immune evasion. Here, we identified 3β-hydroxy-5-cholenoic acid (3β-OH-5-Chln) as a cell-active inhibitor of SULT2B1b. 3β-OH-5-Chln inhibited the cholesterol sulfotransferase activity of SULT2B1b in vitro and suppressed CS production from cancer cells expressing SULT2B1b. In vivo administration of 3β-OH-5-Chln locally reduced CS level in murine CS-producing tumors and increased infiltration of CD8+ T cells. When combined with immune checkpoint blockade or antigen-specific T cell transfer, 3β-OH-5-Chln suppressed the growth of CS-producing tumors. These results demonstrate that pharmacological inhibition of SULT2B1b can promote antitumor immunity through suppressing CS-mediated T cell exclusion.
https://researchmap.jp/-kunimura-/published_papers/36729302