Front. Immunol. 2025 (Koutsogiannaki et al.)
DOCK2 as a novel CD11c ligand in neutrophils to regulate reactive oxygen species production
Koutsogiannaki S, Hou L, Alhamdan F, Mastali M, Murray C, Van Eyk J, Kunimura K, Yuki K:
Frontiers in Immunology 16: 1692451, 2025.
Abstract:
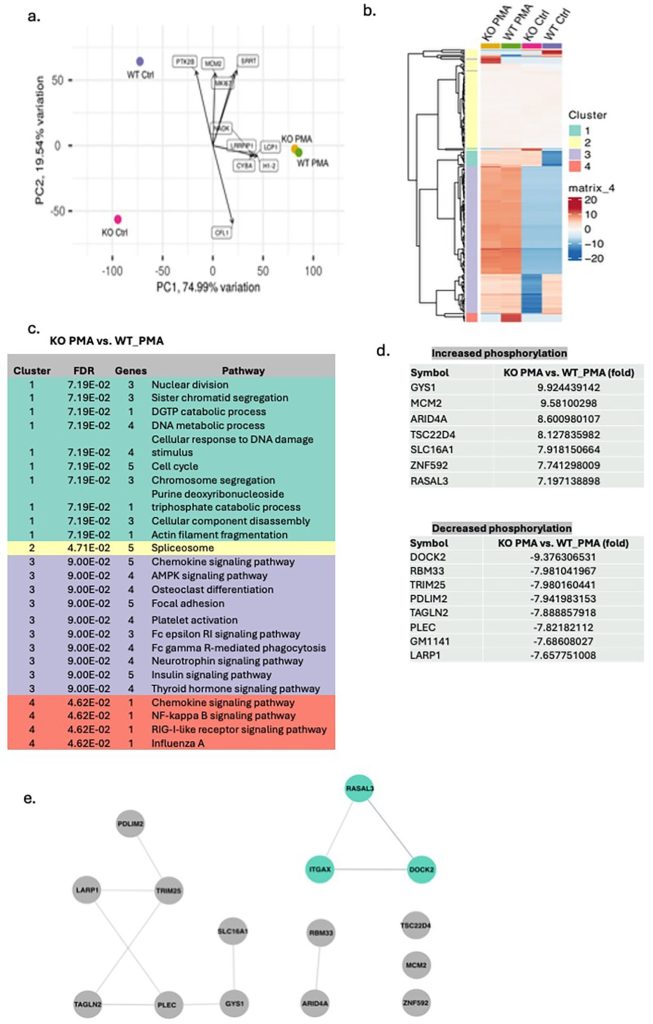
CD11c (integrin αX) is one of the β₂ integrin members traditionally recognized as a dendritic cell marker. It forms the CD11c/CD18 heterodimer—also known as complement receptor 4 (CR4)—and mediates ligand binding to complement fragments, fibrinogen, and intercellular adhesion molecules in vitro. Although historically its expression on dendritic cells and a subset of macrophage populations has been well recognized, recent findings reveal that it demonstrates broader expression profile, including in neutrophils. In neutrophils, CD11c is predominantly intracellular, suggesting a non-canonical role beyond cellular adhesion. We previously identified IQGAP1 as an intracellular binding partner of CD11c/CD18, implicating this interaction in neutrophil maturation. Here mature CD11c-deficient neutrophils displayed impaired reactive oxygen species (ROS) generation while maintaining normal phagocytosis, indicating a selective defect in oxidative burst. Given the central role of NADPH oxidase and Rac activation in ROS production, we hypothesized that CD11c would influence this pathway. Phospho-proteomic profiling revealed reduced phosphorylation of the Rac guanine nucleotide exchange factor DOCK2 in CD11c-deficient neutrophils upon phorbol 12-myristate 13-acetate (PMA) stimulation. The analysis involving immunoprecipitation and proteomics confirmed a CD11c–DOCK2 association. These results supported a model in which CD11c would directly engage DOCK2 to promote Rac activation and NADPH oxidase function, uncovering a novel integrin-mediated mechanism regulating neutrophil effector activity. This work expands the functional repertoire of CD11c and provides a new insight into integrin signaling in innate immunity.
https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2025.1692451/full